 All isotopes of polonium (Po), the only metal in group 16, are radioactive, and only one element in the group, tellurium (Te), can even be described as a semimetal. 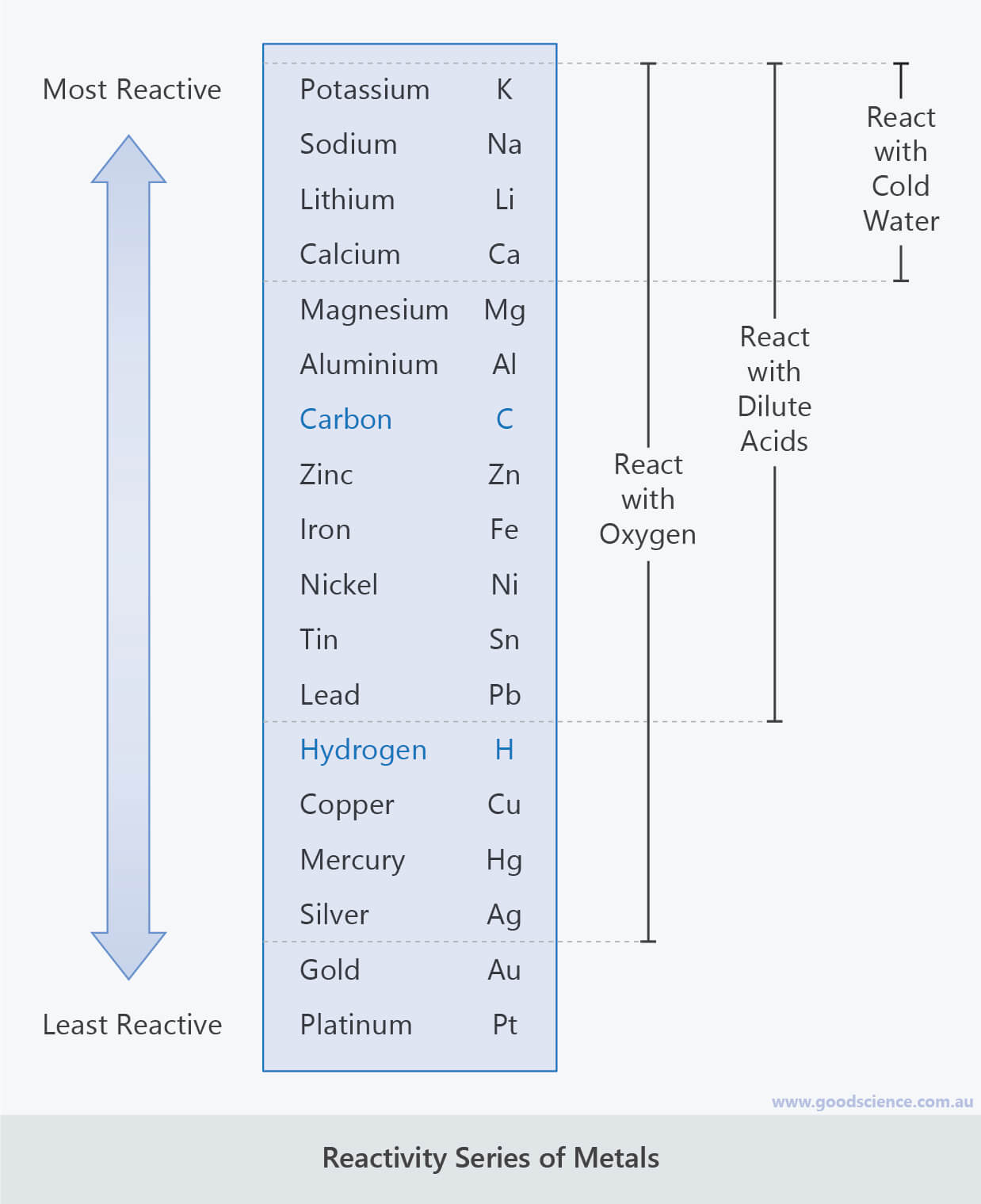
The chalcogens are the first group in the p block to have no stable metallic elements. Most reactive metals are at the top while the least reactive metals at the bottom. To understand the trends in properties and reactivity of the group 16 elements: the chalcogens. Learn how reactivity works, how it is influenced by the periodic table, and how it differs from stability and rate. 
The 2+ ions of the alkaline earth metals have a noble gas like electron configuration and are thus form colorless or white compounds (unless the anion is itself colored). Reactivity series is a list of metals arranged in decreasing order of their reactivity. Reactivity is a measure of how readily a substance undergoes a chemical reaction, depending on its temperature, valence, and electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
